Trying to make some 3-triethylsilylpyridine to use as a standard for our reactions. Reaction seems straight forward enough.
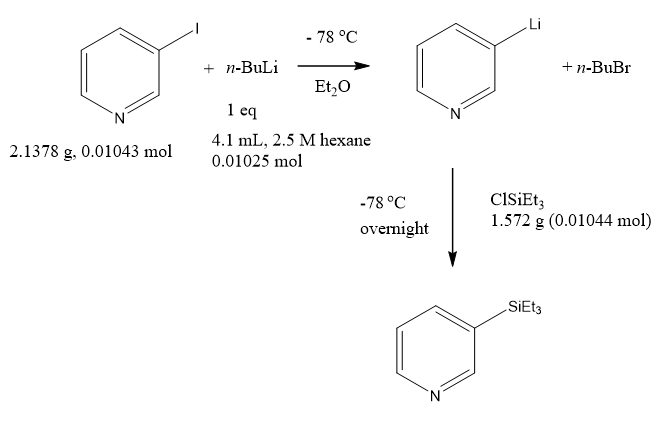
Get dry ice from local Hy-Vee (the one where Kurt Warner used to work). Cool stuff down. Looks like orange crush after the silane addition.

Crude NMR spectrum looked fairly messy.
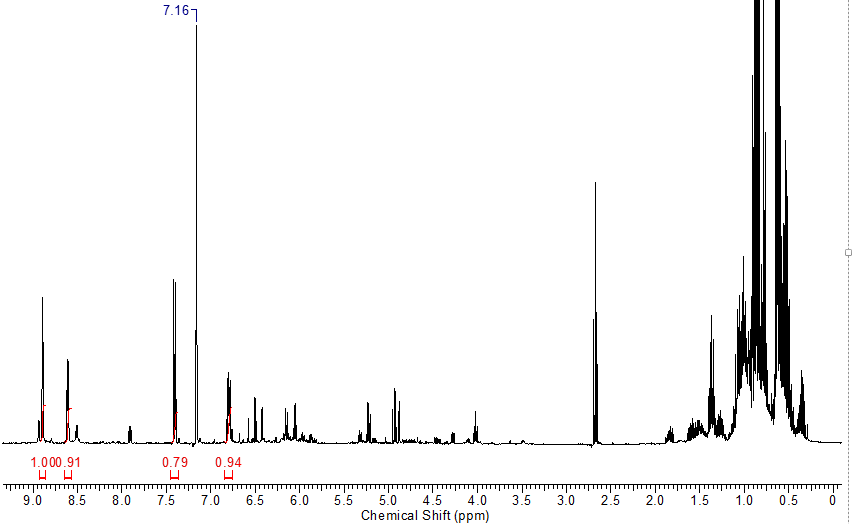
Tried a distillation to clean it up.

Not the best. Still a bunch of silanol.
Another distillation. A real on this time

NMR spectrum says cleanish, the GCMS says otherwise
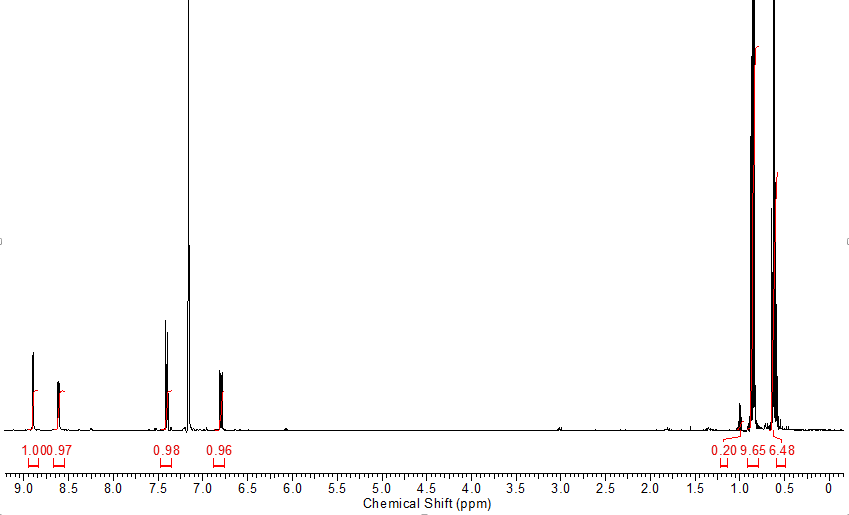
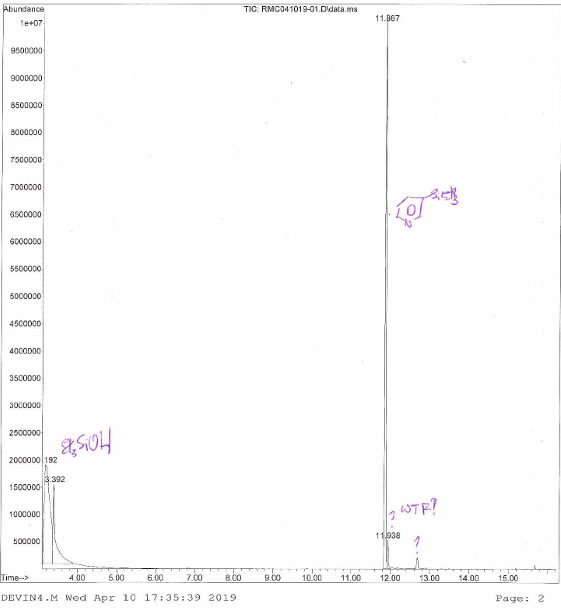
Had to dig deep to clean this thing up. The old prep TLC. The best way to do chromatography. It is like the Hawaiian pizza of chromatography. The absolute best.

Only 66.6 mgs of product. But at least it was clean. And the final mass seems like a sign that I should not pursue this any further.